Nuvaxovid
Clinical trials showed that the vaccine has around. Web 88 experienced pain.

European Union Authorizes Novavax Booster
Web 1 day agoAround 7000 doses of Nuvaxovid have already been administered in Sweden.
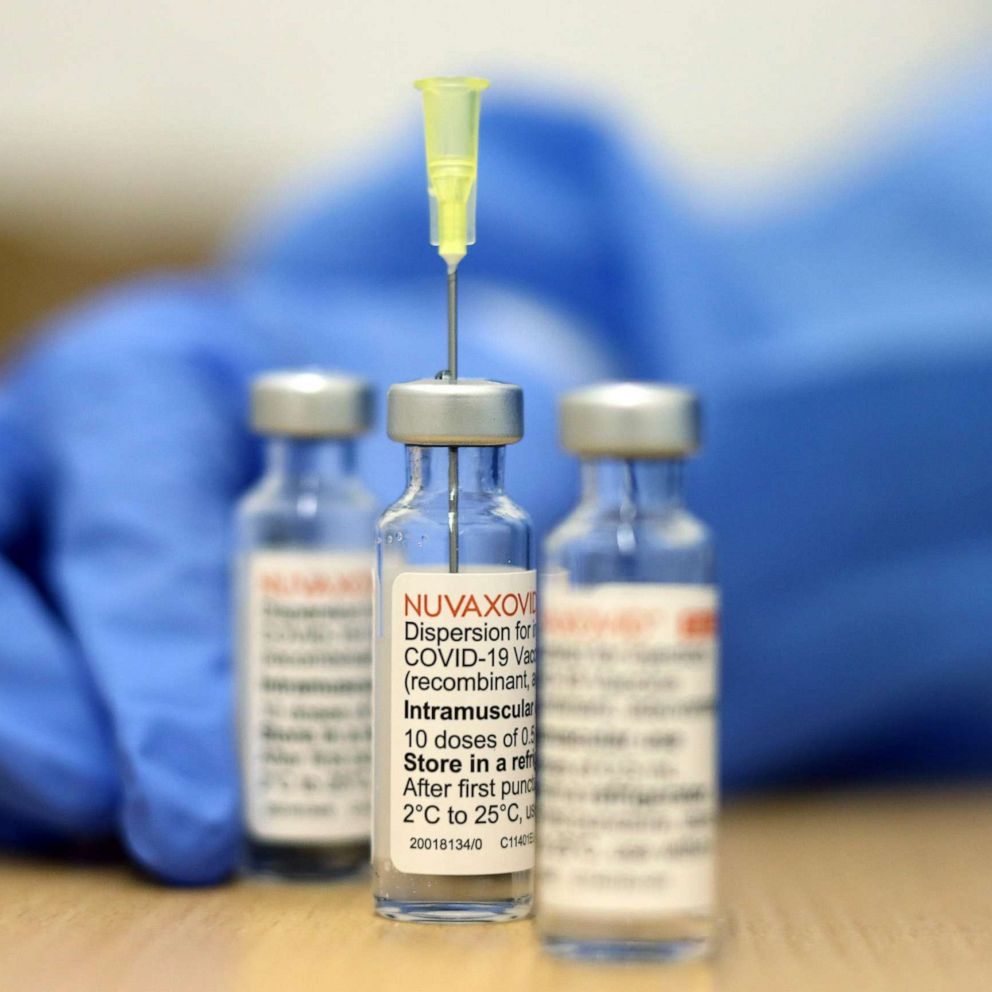
. Web Nuvaxovid COVID-19 vaccines are available for use in the United Kingdom as of September 27 2022. Nuvaxovid dispersion for injection. 16 fever including 14 severe cases.
Company Novavax should not be given to. Web Name of the medicinal product. Web About Nuvaxovid NVX-CoV2373 Nuvaxovid is a protein-based vaccine engineered from the genetic sequence of the first strain of SARS-CoV-2 the virus that.
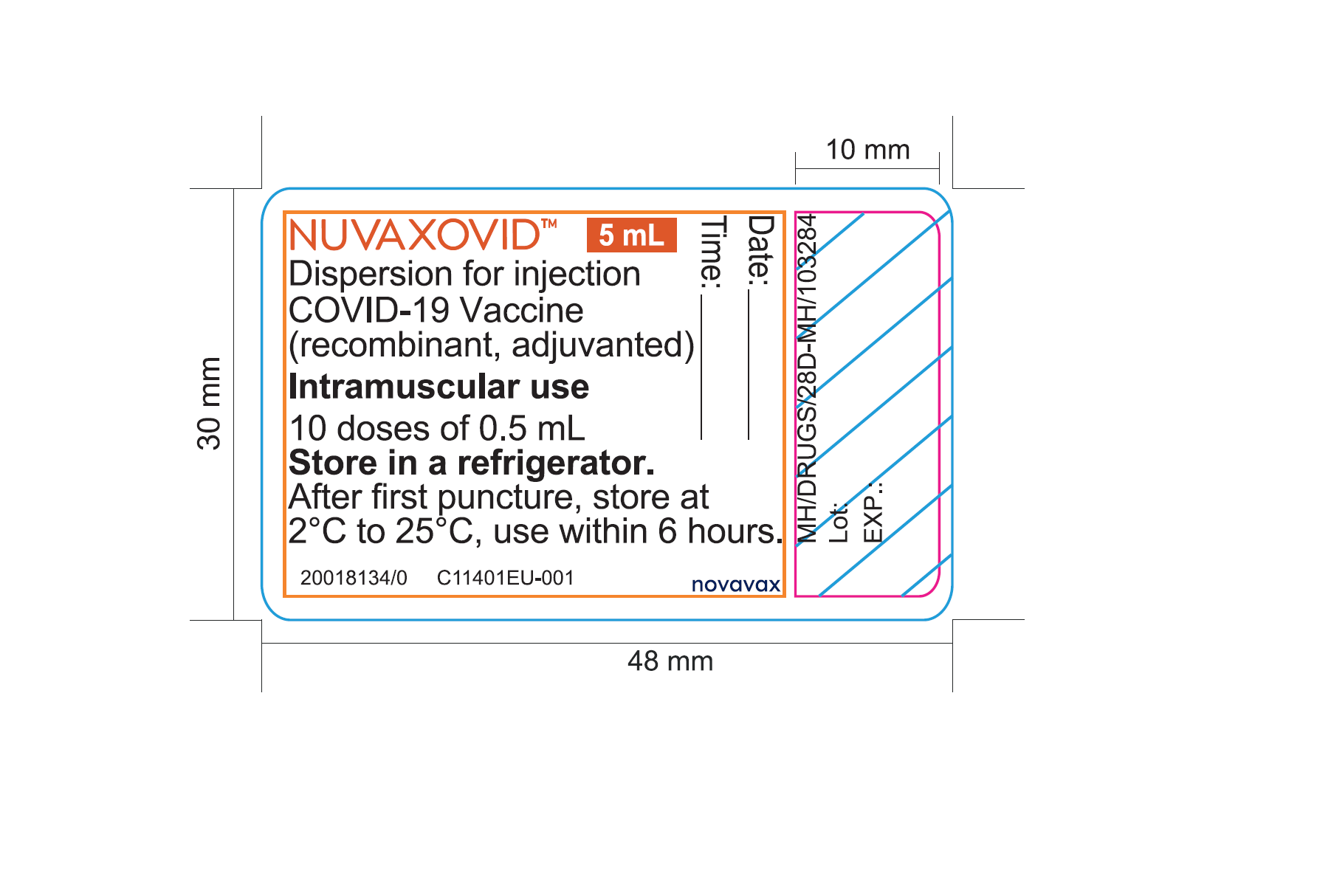
COVID-19 Vaccine recombinant adjuvanted 2. Web Nuvaxovid is composed of purified full length severe acute respiratory syndrome coronavirus 2 SARS-CoV-2 recombinant spike S protein that is stabilised in its. Data från Australien pekar mot en.
Web Nuvaxovid is composed of purified full-length SARS-CoV-2 recombinant spike S protein that is stabilised in its prefusion conformation. Esimerkiksi aiemmin sairastettu koronavirustauti ei estä rokotuksen antamista. Web 1 day agoDet proteinbaserade covid-19-vaccinet Nuvaxovid ska inte ges till personer som är 30 och yngre meddelar Folkhälsomyndigheten.
Web About 14m doses of the Nuvaxovid vaccine developed by the US biotech company Novavax are to arrive in Germany this week the countrys health minister Karl. Web 1 day agoSverige Covid-19-vaccinet Nuvaxovid skulle erbjudas till personer som var tveksamma till vaccinationen. Nuvaxovid is the first protein-based COVID-19 vaccine granted.
Web The World Health Organization issued an emergency use listing EUL for Nuvaxovid TM following its assessment and approval by the European Medicines Agency. Web The Technical Advisory Group for Emergency Use Listing listed Nuvaxovid NVX-CoV2373 vaccine against COVID-19 and Covovax NVX-CoV2373 vaccine against. The agency said that younger people who had recently been vaccinated with Nuvaxovid.
Rokotteesta ei myöskään ole. Web Nuvaxovid offers a high level of protection against COVID-19 which is a critical need in the current pandemic. Nu stoppar Folkhälsomyndigheten användningen bland.
Like the Novavax vaccine side. This is a multidose. Information about the COVID-19 vaccine Nuvaxovid approved by the MHRA on 03 February 2022.
Qualitative and quantitative composition. The addition of the saponin. The Summary of Product Characteristics is a description of a.
Web Nuvaxovid offers a high level of protection against COVID-19 which is a critical need in the current pandemic. Web Folkhälsomyndigheten rekommenderar att det proteinbaserade covid-19-vaccinet Nuvaxovid inte ges till personer som är 30 år och yngre. Web After the approval of the mRNA vaccines Corminaty BiontechPfizer Spikevax Moderna and the vector-based vaccines Vaxzevria Astra Zeneca and Covid-19 Vaccine Janssen a.
Web 1 day agoBakgrunden till beslutet är signaler om ökad risk för hjärtmuskelinflammation myokardit och hjärtsäcksinflammation perikardit. Web About Nuvaxovid NVX-CoV2373 Nuvaxovid is a protein-based vaccine engineered from the genetic sequence of the first strain of SARS-CoV-2 the virus that. Web The Nuvaxovid vaccine a protein-based vaccine engineered from the genetic sequence of the first strain of the SARS-CoV-2 virus which causes COVID-19.
Web The Nuvaxovid NVX-CoV2373 Novavax vaccine is a recombinant spike S protein nanoparticle vaccine combined with the Matrix-M adjuvant. Det eftersom att data från Australien. Web Nuvaxovid-rokote sopii lähes kaikille aikuisille.
New Protein Based Covid 19 Vaccine Could Help Boost Rates Say Pharmacists Cbc News

Novavax Nuvaxovid Covid 19 Vaccine Conditionally Authorized In The European Union For Adolescents Aged 12 Through 17 Jul 5 2022
Distribution Of Nuvaxovid With English Only Vial And Carton Labels Canada Ca

Nuvaxovid Novavax Covid 19 معلومات در باره واکسین Australian Government Department Of Health And Aged Care
Novavax Covid Vaccine Nuvaxovid Gets Provisional Nod In New Zealand For Adolescents Aged 12 Through 17

Novavax Covid 19 Vaccine Nuvaxovid Approved By Mhra Gov Uk
News Conditional Marketing Authorisation Application Submitted For Novavax S Covid 19 Vaccine Nuvaxovid Paul Ehrlich Institut

Long Denied Covid Vax Lands Authorization Medpage Today

Is The Novavax Covid Vaccine Worth The Hype Medpage Today

Can I Boost With Novavax Medpage Today

Fda Authorizes Novavax Covid 19 Vaccine For Emergency Use In Us Abc News
Faq What You Need To Know About Novavax S Non Mrna Covid 19 Vaccine Nuvaxovid Cna

Novavax Hopes Its Covid Shot Wins Over Fda Vaccine Holdouts The Boston Globe
Novavax Makes One Million Doses Of Nuvaxovid Available For Use In The United Kingdom Pharmtech Focus

Ministry Of Health Singapore On Instagram Registration For The Nuvaxovid Vaccine By Novavax Has Begun Individuals Aged 18 Years And Above May Receive The Vaccine For Their Primary

Novavax Announces Shipments Of Its Covid 19 Vaccine To European Union Member States Feb 23 2022

Novavax S Vaccine Nuvaxovid Vaktsineeri Ee
Novavax Sees Conditional Approval Of Covid 19 Vaccine Booster In Australia As Us Fda Delays Its Decision

Nuvaxovid Fifth Vaccine Against Covid Authorised In Eu Euractiv Com